Coumarins
| Sections |
| Structure |
| Structure - Activity Relationships |
| Pharmacology |
| Biochemical Mechanism of Action |
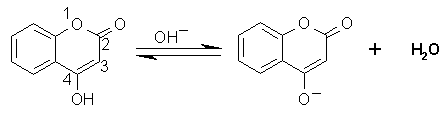
Coumarin and its derivatives are principal oral anticoagulants. Coumarin is water insoluble, however 4-hydroxy substitution confers weakly acidic properties to the molecule that makes it water soluble under slightly alkaline conditions (Equation below).

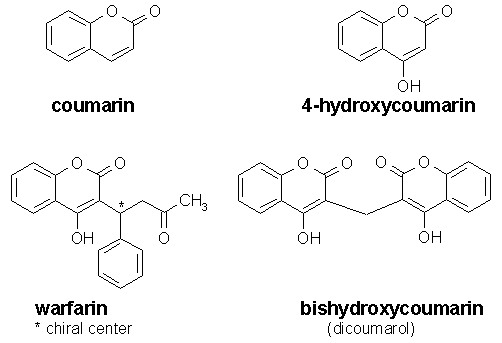
The structures of coumarin and its derivatives are as shown below. Warfarin is marketed as the sodium salt. It has one chiral center. The S(-) isomer is about 5 - 8 times more potent than the R(+) isomer, however, commercial warfarin is a racemic mixture. Click on the appropriate hyperlinks to visualize the three-dimensional structures of individual coumarins.
Structure - Activity Relationships
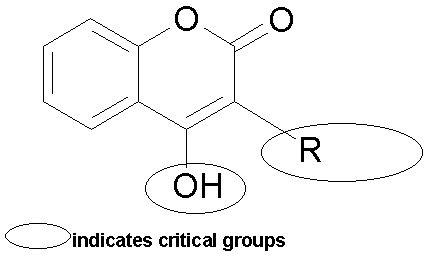
Coumarin and 4-hydroxycoumarin do not possess anticoagulant activity. Link, who pioneered the isolation and characterization of bihydroxycoumarin (dicoumarol) from sweet clover, concluded that the minimal requirements for anticoagulant activity are 4-hydroxy group, a 3-substituent, and a bis molecule (see below).
Coumarins exert their effect in vivo only after a latent period of 12 to 4 hours and their effect lasts for 1.5 to 5 days. The observed slow onset may be due to the time required to decrease predrug prothrombin blood levels, whereas the long duration of action observed with warfarin may be due to the lag time required for the liver to resynthesize prothrombin to predrug blood levels.
Coumarins and 1,3-indandiones (later section) interact with certain drugs. For example, the action of oral anticoagulants can be enhanced by drugs such as phyenylbutazone and salicylates while antagonized by barbiturates and vitamin K.
Biochemical Mechanism of Action
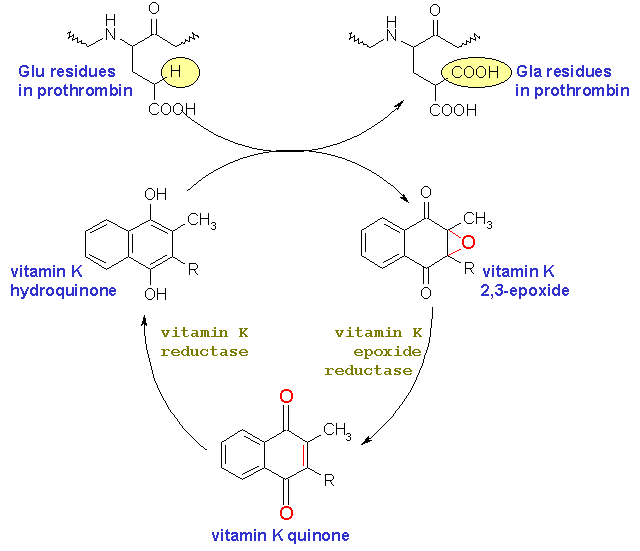
Coumarins are competitive inhibitors of vitamin K in the biosynthesis of prothrombin.
The coagulation cascade relies on the conversion of prothrombin to thrombin in a very important step. However, this conversion depends on the presence of 10 g-carboxyglutamic acid (GLA) residues in the N-terminus of prothrombin. The multiple Gla residues form a binding site for Ca+2. Under normal circumstances 10 glutamic acid (Glu) residues of prothrombin are converted to Gla residues in a post-translational modification.
This post-translation modification is catalyzed by an enzymes vitamin K reductase and vitamin K epoxide reductase. Vitamin K is a co-factor in this conversion reaction. Thus it cycles between a reduced form and an epoxide form. Because of their structural similarity with vitamin K coumarins are thought to bind the enzymes, vitamin K reductase and vitamin K epoxide reductase, without facilitating the conversion of Glu residues of prothrombin to Gla. Thus prothrombin cannot be acted upon by factor Xa.
[top] [session home] [home] [department of medicinal chemistry] [school of pharmacy]
©2000 VCU School of Pharmacy
Revised: January 5, 2000
Questions or Comments : Dr. Umesh R. Desai